Número Atual: Julho-Setembro 2019 - Volume 3 - Número 3
- Imprimir
- Indicar
- Estatísticas
- (0)
Comentários - Como Citar
- Download da Citação
- Artigos Relacionados
-
Outros dos
Autores
Artigo Original
Dispositivos intranasais de medicamentos são uma potencial fonte de contaminação das vias aéreas
Intranasal drug delivery devices are a potential contamination source of airways
Felipe Maciel Pereira1; Lidiane de Castro Soares2; Isadora Passamani Reis Innocencio3; Luísa Costa Oliveira Valente1; Beatriz Julião Vieira Aarestrup3,4; Fernando Monteiro Aarestrup3; Akinori Cardozo Nagato3,5
DOI: 10.5935/2526-5393.20190042
1. Vassouras University, Laboratory of Microbiology - Vassouras, RJ, Brazil
2. Federal Rural University of Rio de Janeiro, Department of Veterinary Microbiology and Immunology - Rio de Janeiro, RJ, Brazil
3. Federal University of Juiz de Fora, Laboratory of Immunopathology and Experimental Pathology - Center for Reproductive Biology - CRB - Juiz de Fora, MG, Brazil
4. Federal University of Juiz de Fora, Hystology Department - Juiz de Fora, MG, Brazil
5. Federal University of Juiz de Fora, Physiology Department - Juiz de Fora - Minas Gerais - Brasil
Endereço para correspondência:
Akinori Cardozo Nagato
E-mail: akynory@hotmail.com
No conflicts of interest declared concerning the publication of this article.
Submitted: 03/06/2019
Accepted: 03/18/2019
RESUMO
BACKGROUND: The use of intranasal drug delivery devices (IDDD) for the treatment of allergic rhinitis (AR) is frequent because they are simple, efficient, and safe, and mainly because they are perceived as low-risk. However, it is speculated that contact between the nasal mucosa and an IDDD may give rise to infections once the nose is colonized by bacteria, and there are currently no proper instructions for IDDD sanitization. The objective of this study was to evaluate the possibility of contamination of an IDDD for topical medication after simulating use in healthy individuals.
METHODS: The in vitro study consisted of 14 healthy individuals of both sexes, between the ages of 18 and 24 years. Samples were collected immediately after the opening of each IDDD and after simulating use by the subjects. Afterwards, the samples were deposited in tubes and kept in an incubator at 37 °C. After 48 hours, the samples were inoculated on Müller-Hinton agar. Qualitative analyses of the appearance of the samples were performed after 24 and 48 hours, and after 72 hours the presence or absence of bacteria was evaluated macroscopically.
RESULTS: After 24 hours of incubation, 21.4% (n = 3) of the samples presented with a turbid appearance and after 48h, 71% (n = 10) of the samples presented with a turbid appearance and positive bacterial growth.
CONCLUSION: The results suggest that IDDDs for topical medications may be important sources of contamination or recontamination of the nasal mucosa of individuals who are being treated for upper respiratory tract conditions. A better understanding of the risks of re-using IDDDs after previous contact with the nasal mucosa will improve guidelines on hygiene procedures and prevention of related risks.
Descritores: Administration, intranasal, nasal mucosa, contamination, bacteria.
Introduction
Nasal congestion (the perception of reduced airflow or a feeling of fullness in the nose) is caused by a variety of environmental factors and diseases, including allergic rhinitis (AR). According to the International Phase III Study of Asthma and Allergies in Childhood (ISAAC), AR affects 14.9% of individuals aged 6-7 years and 39.7% between 13-14 years worldwide.1 Allergic rhinitis usually causes nasal obstruction, a condition frequently perceived and described by patients,2 and leads to self-medication.3
The nose provides easy access for delivery of medications and vaccines,4 although the nasal vestibule is highly colonized and presents a potential risk for infection.5 Nasal decongestants are among the most accessible and most frequently used treatments6 and have high rates of self-medication in Brazil.7,8 In general, they are widely sought due to their rapid efficiency in improving airflow through the nasal cavity8 and generate a sense of well-being mainly in those individuals with a feeling of nasal congestion.2,9 Usually this occurs in infections of the upper airways, as in allergic rhinitis, which affects about 29.6% of adolescents in Brazil.10
The nasal microbiota, even under physiological conditions, commonly presents bacteria such as Staphylococcus aureus, Enterobacter aerogenes, Klebsiella pneumoniae, and Pseudomonas aeruginosa.11 It has been reported that nasal colonization by Staphylococcus aureus affects 20-30% of healthy individuals.12 However, because of a weakened immune response, a pharmacologically suppressed immune system, or nasal mucosal contact with bacteria not recognized by the immune system ('not self', in immunology), bacterial proliferation is favored, allergic or non-allergic immune responses are activated, and inflammatory mediators like histamine and prostaglandins are released. This induces inflammatory cell recruitment, vasodilation, increased local permeability, and activation of goblet cells. These actions can lead to signs and symptoms such as edema, pruritus, nasal congestion, rhinorrhea, coryza, partial or total obstruction of airflow, and dyspnea.13,14
For the adjuvant treatment of these conditions, in order to promote mucociliary cleaning (which removes crystallized material from the nasal cavity), decrease the viscosity of the nasal mucus, and allow the return of airflow, hypertonic solutions (NaCl solutions), nasal decongestants (aromatic amines, aliphatic amines, and imidazole derivatives) and nasal topical corticosteroids (beclomethasone, budesonide, fluticasone propionate, mometasone, triamcinolone, fluticasone furoate, ciclesonide)15 are frequently prescribed.
Topical medications with intranasal drug delivery devices (IDDD) have been widely indicated as possible therapeutics,16 especially among patients with rhinitis (allergic or not), allergic rhinoconjunctivitis, rhinosinusitis, and nasal polyposis. In the specific case of rhinitis, glucocorticoids have been mentioned as the gold standard in improving patients' quality of life.2,6
IDDDs are frequent modes of treatment in both adults and children, mainly because they are considered a simple, effective, and safe treatment, especially from the point of view of risks related to the possible induction of lesions in the nasal mucosa by direct contact. Saline solutions, for example, have become popular as they are inexpensive, practical, and well-tolerated, although more studies are required.8,17,18
On the other hand, from the microbiological point of view, it is believed that IDDDs may not be safe, especially since during drug or saline solution delivery in domestic environments, direct contact between the face of the IDDD and the nasal mucosa can occur, which risks contamination and/or recontamination in the individual. To prevent this from happening, it is necessary that the user be properly instructed in relation to maintaining hygiene with the IDDD making direct contact with the nasal mucosa. However, these guidelines are scarce in the literature.
Taking this into account, the present study evaluated the possibility of contamination of an IDDD after simulating use in healthy individuals, with the intention of alerting the scientific community about the possible recontamination of the nasal mucosa of individuals being treated for conditions of the upper respiratory tract.
Materials and Methods
The in vitro experimental study investigated the possible contamination of an IDDD after single simulated contact with the nasal vestibule of healthy individuals. The study was carried out at the Laboratory of Microbiology and Biomorphology and Experimental Pathology of the University of Vassouras (Vassouras, Rio de Janeiro, Brazil) and the Laboratory of Immunopathology and Experimental Pathology at the Center for Reproductive Biology (CRB; Federal University of Juiz de Fora, Juiz de Fora, Minas Gerais, Brazil).
Subjects
A convenience sample was used in this study. We chose 14 healthy individuals, 12 men and 2 women aged between 18 and 24, who had no anatomical nasal abnormalities, no diagnosis of sinusitis, and no recurrent nasal bacterial infection or purulent nasal discharge. At the time of the study, none of the subjects were taking antibiotics. This information was collected from a semi-structured questionnaire specifically developed for this study. All individuals were instructed about all stages of the study and voluntarily signed a consent form. The study was carried out with the approval of the ethics committee (No. 1,596,547), according Resolution No. 466/12 of the National Health Council.
Sampling
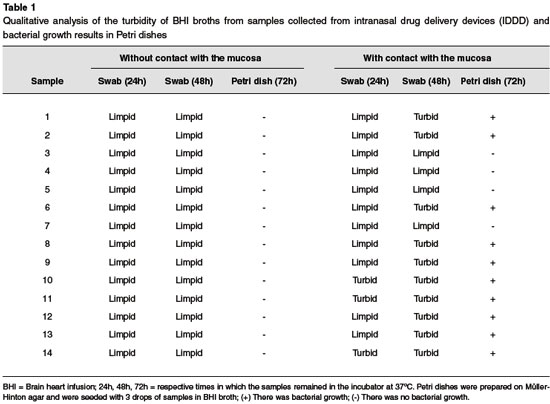
Sealed packages containing a nasal solution of banzalconium chloride, sodium chloride, and nafazoline hydrochloride (Neosoro®; Neo Química, Rio de Janeiro) were opened 10 cm away from a Bunsen burner flame, while handling the bottles with a sterile glove. Immediately after opening each IDDD, an initial sample from the entire surface of the IDDD was collected with a sterile swab, soaked in 0.9% NaCl solution. After collection, the swab was immediately submerged in brain heart infusion broth (BHI). All subjects were instructed to insert the IDDD in the nasal ostium towards the limen nasi, and to maintain that position for 5 seconds without ejecting the medication, simulating the proper positioning of the device during medication use. Additionally, a second sample was collected following the same criteria mentioned for the swab collection of the first sample. A total of 28 samples (14 without contact with the nasal mucosa and 14 after contact with the nasal mucosa) were collected and incubated at 37ºC for 24 hours. After the samples were taken from the incubator, a first analysis of the culture broth appearance was carried out in order to identify the translucency of the broth and qualitatively examine the turbidity. The samples were then returned to the incubator at 37°C for a further 24 hours, and a second turbidity analysis of the broth was performed (Figure 1).
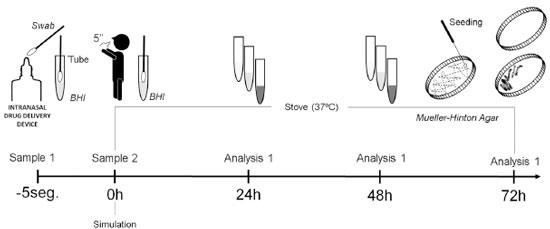
Figure 1 Experimental timeline. BHI - Brain heart infusion. '-5seg', '0h', '24h', '48h' e '72h' - respective times (in hours, "h", or seconds, "sec") in which each step was performed. '1ª collect' - At the beginning of the experiment (-5sec), intranasal drug delivery device (IDDD) samples were collected with sterile Swab, immediately after opening the package, and immersed in a tube containing BHI broth and kept in an incubator at 37ºC. '2ª collect' - Next (0h), IN samples were collected with sterile Swab immediately after the simulation of the IDDD use and immersed in a tube containing BHI broth and kept in an incubator at 37ºC. '1ª analyze' e '2ª analyze' - qualitative analysis of the turbidity of BHI broths. Within 48h the samples were seeded from Petri dish in culture media (Müller-Hinton agar). '3ª analyze' - qualitative analysis of bacterial growth
Furthermore, the samples grown in BHI broth were seeded in Müller-Hinton agar and incubated for 24 h at 37°C. After this period the plates were investigated for the presence or absence of bacterial growth. The plates were placed on a flat surface, kept at a fixed distance of 30 cm, and photographed (NIKON L810, 10 MP, 26x optical zoom, 4x digital zoom, 4608 x 3456 resolution, ISO sensitivity 80-1600) on a tripod in the same lighting conditions.
A descriptive analysis of 20 package inserts of nasal topical medicines registered in the National Agency of Sanitary Surveillance's (ANVISA) electronic system was carried out in order to identify information on: (1) guidelines on the correct positioning of an IDDD; (2) recommendations for IDDD hygiene; and (3) possible contamination of an IDDD.
Results
The BHI broth from the samples collected immediately after opening the intranasal drug delivery device without contact with the mucosa had a clear appearance after both 24h and 48h in culture, suggesting no bacterial growth. This result was later confirmed when the seeded samples did not demonstrate bacterial growth in a Petri dish after 72h.
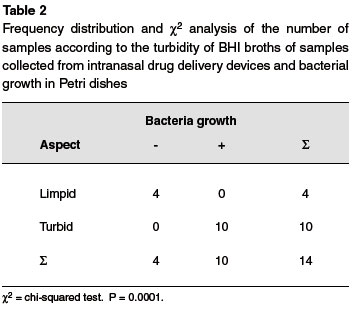
From the samples that had contact with the nasal mucosa, turbidity was observed in the first 24 hours in 21.4% (n = 3) of samples. After 48h, this result was even higher, where 71.4% (n = 10) of samples had a turbid appearance. This result was also confirmed by bacterial growth on Müller-Hinton agar. (Chart 1e, Figure 2).
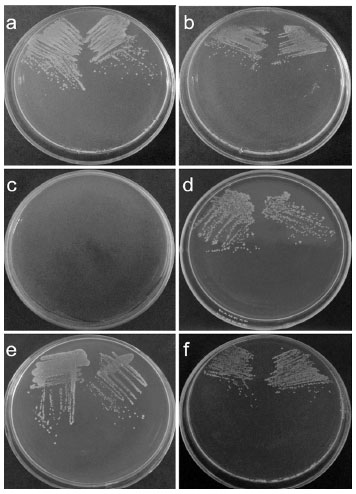
Figure 2 Petri dish prepared with Müller-Hinton agar culture medium were seeded from the 3 drops of samples collected after the simulation of the use of the intranasal drug delivery device in BHI liquid. Plates a, b, d, e and f resulted in visible bacterial growth
Twenty percent of the package inserts analyzed in the present study did not provide information on the positioning of IDDDs in relation to the patients' nostrils when describing recommendations for drug delivery. Although most package inserts had some recommendations regarding IDDD hygiene, 25% of them had no clear recommendations. Among the package inserts with no hygiene recommendations, there was at least a suggestion that IDDDs should not be shared in order to avoid contamination (Chart 3).
Discussion
In the current study, we tested the possible risk of contamination of IDDDs used in the treatment of upper respiratory tract diseases, as during application it can come in direct contact with the nasal mucosa, and this contact can be an important area of reinfection of the nasal cavity while re-using the IDDD.
We investigated the possibility of contamination because the guidelines on the correct positioning of IDDDs and hygienic procedures related to nasal instillation are unclear. We have shown, after simulating the use of IDDDs, a high risk of contamination among healthy individuals and confirmed inconsistencies in hygiene methods on the package inserts of these drugs.
Topically applied nasal drugs for patients with upper respiratory tract diseases are frequently prescribed8 and they are clinically required for facilitating the delivery of the active compound because the nasal vestibule is widely vascularized and presents a large surface area.19 On the other hand, in order to achieve maximum efficiency, the pharmaceutical industry recommends regular use of topically applied nasal drugs20 and there are reports that their efficacy depends on daily use.21
In fact, there is no consensus regarding the dosage or duration of treatment with topical nasal decongestants, including those that can be purchased without a prescription (except for those containing vasoconstrictors).22 These factors may result in an increase in the number of times the IDDD contacts the nasal vestibule and raises the possibility of contamination and/or recontamination, increasing the risk to users.
Another factor that increases the possibility of contamination and/or recontamination is advice from pharmacists without adequate knowledge of the indications, contraindications, dosage, adverse effects, and possible drug interactions for the treatment of diseases - a fact routinely observed in Brazil in relation to respiratory diseases (Balbani, Sanchez, Butugan, 1996; Balbani et al., 1996).
There are no clear reports of risks associated directly with IDDDs. There is some evidence, however, that shows possible IDDD contamination in the postoperative period of endoscopic surgery or the contamination of the liquid when inside the patient.23
Concerns about the risks associated with the use of nasal topical medications are generally not related to the hygiene procedures employed while handling and using nasal applicators. There are few reports on the need to sanitize the nasal vestibule as an adjuvant treatment.21 Usually, the highlighted risks are those related to adverse effects of topical nasal medications, such as reduced milk production during breastfeeding,24 risk of hypotension and bradycardia in elderly patients,25 local irritation, bleeding, septal perforation, hypothalamic-pituitary-adrenal axis interference, ocular effects, growth effects, bone resorption, and cutaneous effects.10
Self-medication and abuse of nasal decongestants has been identified as an important risk factor due to drug-induced rhinitis by a rebound reaction.26 In the literature, other reports point to an impairment of mucociliary clearance by the use of topical nasal medications.27 In this case, impaired mucociliary dynamics lead to a favorable microenvironment for bacterial growth, increasing the possibility of IDDD contamination during direct contact with the nasal mucosa.
The present study has also shown that the package inserts of the analyzed drugs are not clear regarding the specific positioning of an IDDD. Concerns about the clarity of health descriptions, such as labeling and packaging patterns, have strong implications for therapeutic efficacy and patient safety, especially when such information comes from the internet.28
In general, studies that focus on investigating the relationship between the diameter, depth, and angle of the nasal spray position in relation to the anatomy of the airways restrict themselves to discussing only the information about the dose reached in the therapies,4 but there are no reports of the possibility of contamination and/or recontamination of the applicator. New studies could clarify if ergonomic aspects can interfere in the contact area between an IDDD and the nasal mucosa, and consequently in the possibility of contamination and/or recontamination of the IDDD.
The risk of contamination was indicated in 75% of package inserts analyzed in the present study, both when considering the severity of the sharing of IDDD among users and when considering hygiene recommendations, although the information on the hygiene methods used is not clear. Therefore, there is no way to know if proper hygiene procedures are actually performed by the patients. Normally, when information is unclear and/or there is a low perception of risk by users, there is a lower tendency towards adherence to hygiene methods.29
Topical nasal medications are among the most common medications associated with self-medication3. It is known, however, that self-medication increases risk for the patient, and may have significant side effects.30 In the present study, it was demonstrated that IDDD contamination is not easily predicted, since it requires specific culture techniques to identify bacterial growth. As could be observed, even with the guided use of IDDD by healthy individuals, there was a risk of contamination after a single simple simulation of the use of the nasal applicator and thus should be considered an important focus of possible recontamination of the upper airways.
In the present study, we were neither able to identify the bacterial types in vitro nor evaluate the rate of contamination of the IDDDs in contact with the nasal mucosa of individuals with previous upper respiratory tract infections. It is speculated that if an IDDD made contact with previously infected nasal mucosa, it could significantly increase the risk of contamination, although one study has shown that the variation of the microbiota between individuals was more important than the disease state.11 If this latter point is considered on a large scale, better guidance on IDDD hygiene as well as guidance on how to re-use IDDDs should be treated as a public health problem, and this study suggests that preventive measures should be employed.
Environmental factors can affect the nasal microbiome, with potential effects on an allergic subject's health, resulting in a decrease in quality of life and increased nasal symptoms. Research implications suggest a new paradigm of hygiene for IDDDs. Whether IDDDs can alter the nasal microbiome or affect the course of inflammation in bacterial rhinosinusitis remains to be seen, and current therapeutic methods may need to be modified. However, further studies will be needed to clarify these issues.
Conclusion
Our results suggest that IDDDs for topical medications may be considered important sources of contamination/recontamination of the nasal mucosa of individuals who are being treated for conditions of the upper respiratory tract. A better understanding of the risks of re-using IDDDs will help determine guidelines on hygiene procedures and prevention of related risks. Therefore, we recommend that hygiene and disinfection measures should be clearly patientoriented, and that the use of nasal devices should be for individual use only.
References
1. Strachan D, Sibbald B, Weiland S, Ait-Khaled N, Anabwani G, Anderson HR, et al. Worldwide variations in prevalence of symptoms of allergic rhinoconjunctivitis in children: the International Study of AsthmaandAllergiesinChildhood (ISAAC). Pediatr AllergyImmunol. 1997 Nov;8(4):161-76.
2. Stewart M, Ferguson B, Fromer L. Epidemiology and burden of nasal congestion. Int J Gen Med. 2010;3:37-45.
3. Mehuys E, Gevaert P, Brusselle G, Van Hees T, Adriaens E, Christiaens T, et al. Self-medication in persistent rhinitis: overuse of decongestants in half of the patients. The journal of allergy and clinical immunology In practice. 2014;2(3):313-9. https://doi.org/10.1016/j.jaip.2014.01.009.
4. Djupesland PG. Nasal drug delivery devices: characteristics and performance in a clinical perspective-a review. Drug delivery and translational research. 2013;3(1):42-62 https://doi.org/10.1007/s13346-012-0108-9.
5. Lecomte F, Nouvellon M, Levesque H. Nasal carriage of Staphylococcus aureus. The New England journal of medicine. 2001;344(18):1399-400;author reply 400-1 https://doi.org/10.1056/NEJM200105033441813.
6. Mello Júnior JFd, Mion OdG, Andrade NAd, Anselmo-Lima WT, Stamm AEC, Almeida WLdC, et al. Posicionamento da Academia Brasileira de Rinologia sobre terapias tópicas nasais. Braz j otorhinolaryngol [online]. 2013;79(3):391-400.
7. Arrais PSD, Coelho HLL, Batista MdCDS, Carvalho ML, Righi RE, Arnau JM. Perfil da automedicação no Brasil. Rev Saúde Pública [online]. 1997;31:71-7
8. Lenz D, Cardoso KS, Bitti ACR, Andrade TU. Evaluation of the use of topic nasal decongestants in university students from health sciences courses. Braz J Pharm Sci. 2011;47(4):761-7.
9. Baraniuk JN. Subjective nasal fullness and objective congestion. Proc Am Thorac Soc. 2011;8(1):62-9. https://doi.org/10.1513/pats.201006-042RN.
10. Ibiapina CdC, Sarinho ESC, Camargos PAM, Andrade CRd, Cruz Filho ÁASd. Rinite alérgica:aspectos epidemiológicos, diagnósticos e terapêuticos. J Bras Pneumol.2008;34(4):230-40.
11. Biswas K, Hoggard M, Jain R, Taylor MW, Douglas RG. The nasal microbiota in health and disease: variation within and between subjects. Front Microbiol. 2015;6:134. https://doi.org/10.3389/fmicb.2015.00134.
12. Reinato LAF, Pereira FMV, Lopes LP, Pio DPM, Gir E. Colonização nasalem profissionais de enfermagemdeunidadesespecializadas em HIV/aids. Rev Bras Enferm. 2015;68:320-4.
13. Larche M, Akdis CA, Valenta R. Immunological mechanisms of allergen-specific immunotherapy. Nature Reviews Immunology. 2006;6(10):761-71. https://doi.org/10.1038/nri1934.
14. de Bot CM, Moed H, Bindels PJ, van Wijk RG, Berger MY, de Groot H, et al. Exhaled nitric oxide measures allergy not symptoms in children with allergic rhinitis in primary care: a prospective cross-sectional and longitudinal cohort study. Prim Care Respir J.2013;22(1):44-50. doi: 10.4104/pcrj.2013.00009.
15. Bousquet J, Hellings PW, Agache I, Amat F, Annesi-Maesano I, Ansotegui IJ, et al. Allergic Rhinitis and its Impact on Asthma (ARIA) Phase 4 (2018):Change management in allergic rhinitis and asthma multimorbidity using mobile technology. J Allergy Clin Immunol. 2019 Mar;143(3):864-79. doi: 10.1016/j.jaci.2018.08.049.
16. Jung M, Lee JY, Ryu G, Lee KE, Hong SD, Choi J, et al. Beneficial effect of nasal saline irrigation in children with allergic rhinitis and asthma: A randomized clinical trial. Asian Pac J Allergy Immunol. 2019 Apr 23. doi: 10.12932/AP-070918-0403. [Epub ahead of print].
17. Sakano E, Sarinho ESC, Cruz AA, Pastorino AC, Tamashiro E, Kuschnir F, et al. IV Brazilian Consensus on Rhinitis - an update on allergic rhinitis. Braz j otorhinolaryngol [online]. 2018;84(1):3-14. https://doi.org/10.1016/j.bjorl.2017.10.006.
18. Karatas A. Pretreatment prediction of the outcomes of intranasal steroid sprays in cases with inferior turbinate hypertrophy. Turk Arch Otorhinolaryngol. 2017;55(3):105-10. doi: 10.5152/tao.2017.2443 https://doi.org/10.5152/tao.2017.2443.
19. Bitter C, Suter-Zimmermann K, Surber C. Nasal drug delivery in humans. Curr Probl Dermatol. 2011;40:20-35. doi: 10.1159/000321044.
20. Juniper EF, Guyatt GH, O'Byrne PM, Viveiros M. Aqueous beclomethasone diproprionate nasal spray: regular versus "as required" use in the treatment of seasonal allergic rhinitis. J Allergy Clin Immunol. 1990 Sep;86(3 Pt 1):380-6.
21. Bartle J. How to use a corticosteroid nasal spray. Nurs Stand. 2017;31(52):41-3. doi: 10.7748/ns.2017.e10684.
22. Bucaretchi F, Dragosavac S, Vieira RJ. Exposição aguda a derivados imidazolínicos em crianças. J Pediatr (Rio J). 2003;79(6):519-24.
23. Welch KC, Cohen MB, Doghramji LL, Cohen NA, Chandra RK, Palmer JN, et al. Clinical correlation between irrigation bottle contamination and clinical outcomes in post-functional endoscopic sinus surgery patients. Am J Rhinol Allergy. 2009;23(4):401-4. doi: 10.2500/ajra.2009.23.3338.
24. Chaves RG, Lamounier JA. Uso de medicamentos durante a lactação. J. Pediatr (Rio J). 2004;80(5 suppl):s189-s198.
25. Alshami A, Romero C, Avila A, Varon J. Management of hypertensive crises in the elderly. J Geriatr Cardiol. 2018;15(7):504-12. doi: 10.11909/j.issn.1671-5411.2018.07.007.
26. Yoo JK, Seikaly H, Calhoun KH. Extended use of topical nasal decongestants. Laryngoscope. 1997;107(1):40-3.
27. van de Donk HJ, van den Heuvel AG, Zuidema J, Merkus FW. The effects of nasal drops and their additives on human nasal mucociliary clearance. Rhinology. 1982;20(3):127-37.
28. Veronin M. Packaging and labeling of pharmaceutical products obtained from the internet. J Med Internet Res 2011;13(1):e22. https://doi.org/10.2196/jmir.1441.
29. Piai-Morais TH, Orlandi FdS, Figueiredo RMd. Factors influencing adherence to standard precautions among nursing professionals in psychiatric hospitals. Rev Esc Enferm USP. 2015;49(3):478-85.
30. Servidoni AB, Coelho L, Navarro MdL, Ávila FGd, Mezzalira R. Perfil da automedicação nos pacientes otorrinolaringológicos. Rev Bras Otorrinolaringol. 2006;72:83-8.